Chemistry and Honors Chemistry
The Chemistry curriculum is based on the North Carolina Essential Standards of Chemistry. You can check out what we'll be covering in class below!
Test #1 Content:
Pre-Chemistry:
Chemistry is the study of MATTER.
All matter is made of ATOMS (so it has mass) and it has VOLUME (which takes up space), but how do types of matter differ?
Chemistry is the study of MATTER.
All matter is made of ATOMS (so it has mass) and it has VOLUME (which takes up space), but how do types of matter differ?
Objective 1.1.1: Analyze the structure of atoms, isotopes, and ions.
Using the periodic table, you should be able to take a given atom/isotope/ion and provide the number of protons, neutrons, and electrons for that substance.
Important terms here include: atomic number (number of protons), mass number (number of protons and neutrons), and average atomic mass (decimal number, average of all of the isotopes of that element).
a. Remember, neither atoms nor isotopes have a charge. Ions, however, DO have a charge. This charge will cause a change in the number of electrons when compared to protons. Normal elements have the same number of protons as electrons. Ions will have more or less electrons than protons (depending on the charge).
b. Isotopes all have the same number of protons. However, they have different mass numbers because they have varying numbers of neutrons.
Using the periodic table, you should be able to take a given atom/isotope/ion and provide the number of protons, neutrons, and electrons for that substance.
Important terms here include: atomic number (number of protons), mass number (number of protons and neutrons), and average atomic mass (decimal number, average of all of the isotopes of that element).
a. Remember, neither atoms nor isotopes have a charge. Ions, however, DO have a charge. This charge will cause a change in the number of electrons when compared to protons. Normal elements have the same number of protons as electrons. Ions will have more or less electrons than protons (depending on the charge).
b. Isotopes all have the same number of protons. However, they have different mass numbers because they have varying numbers of neutrons.
In the example above, Calcium (Ca 2+) and Fluoride (F-) are ions. They have the "normal" number or protons and neutrons, but they have more or less electrons compared to the protons, which in normal atoms would be the same number.
Carbon-14, Chlorine-35 and Uranium-235 are all isotopes. Chlorine is the most common isotope, as seen on the periodic table, so the numbers of protons and neutrons are what you would expect. C-14 and U-235 are rarer isotopes, so their number of neutrons differ from the number of neutrons that would be calculated from the most common isotope as seen on the periodic table.
Carbon-14, Chlorine-35 and Uranium-235 are all isotopes. Chlorine is the most common isotope, as seen on the periodic table, so the numbers of protons and neutrons are what you would expect. C-14 and U-235 are rarer isotopes, so their number of neutrons differ from the number of neutrons that would be calculated from the most common isotope as seen on the periodic table.
Calculating the Average Atomic Masses on the Periodic Table:
Remember, the decimal numbers under each (or most) elements on the periodic table are the average atomic mass. To calculate average atomic mass, we multiple the relative abundance of each isotope by the mass number of each isotope. Then, you add the products together.
Sulfur has 4 isotopes, with the following abundances:
S-32 94.93% --> 0.9493 32 x 0.9493 = 30.38
S-33 0.76% --> 0.0076 33 x 0.0076 = 0.25
S-34 4.29% --> 0.0429 34 x 0.0429 = 1.46
S-36 0.02% --> 0.0002 36 x 0.0002 = 0.01 (30.38 + 0.25 + 1.46 + 0.01 = 32.10) Ave. atomic mass = 32.1
Remember, the decimal numbers under each (or most) elements on the periodic table are the average atomic mass. To calculate average atomic mass, we multiple the relative abundance of each isotope by the mass number of each isotope. Then, you add the products together.
Sulfur has 4 isotopes, with the following abundances:
S-32 94.93% --> 0.9493 32 x 0.9493 = 30.38
S-33 0.76% --> 0.0076 33 x 0.0076 = 0.25
S-34 4.29% --> 0.0429 34 x 0.0429 = 1.46
S-36 0.02% --> 0.0002 36 x 0.0002 = 0.01 (30.38 + 0.25 + 1.46 + 0.01 = 32.10) Ave. atomic mass = 32.1
| hchemquiz2.docx | |
| File Size: | 77 kb |
| File Type: | docx |
Objective 1.1.2: Analyze an atom in terms of the location of electrons.
When drawing a Bohr model of an atom, you have some clues based on where the element is located on the periodic table. The period in which you find the element tells you how many rings or energy levels should be present around the nucleus of the atom. The group number reveals how many valence electrons (outside energy level electrons) exist--as long as the element is found in one of the "A" groups (I-VIII A).
When drawing a Bohr model of an atom, you have some clues based on where the element is located on the periodic table. The period in which you find the element tells you how many rings or energy levels should be present around the nucleus of the atom. The group number reveals how many valence electrons (outside energy level electrons) exist--as long as the element is found in one of the "A" groups (I-VIII A).
To perform the quantum version of electron configuration, you follow similar rules. Remember, only 2 electrons (represented by arrows) can fit in each orbital. Orbital families (s, p, d, f) can hold 1, 3, 5, and 7 orbitals respectively. The amount of energy found in electrons increases as each orbital group is added to the electron configuration. "Up" arrows must be filled in on each orbital before the down arrows can be added. When you complete electron configuration, the coefficients represent the ring number and the exponents represent how many electrons are found on that ring.
Remember, the shortcut method can be used for electron configuration if it's a larger element than Krypton. You put the last noble gas (group 18) into brackets and work from there.
Objective 1.1.3: Explain the emission of electromagnetic radiation in spectral form in terms of the Bohr model.
Using your Reference Table, you should be able to explain what happens when electrons are grounded (releasing energy) or excited by absorbing energy. When electrons are pulled closer to the nucleus, energy is emitted (sometimes in the form of visible light, and at other times in the form of invisible energy (infrared radiation, ultraviolet radiation). When electrons absorb energy, they move away from the nucleus of the atom. Any energy that is emitted from electrons is made up of particles called PHOTONS.
a. For example, when electrons move from n = 2 to n =6, the electrons become more excited because they have ABSORBED energy.
b. When electrons move from n = 5 to n = 2, electrons are GROUNDED and energy is RELEASED. Specifically, 434 nanometers of light are emitted, which is visible light. Because it is visible, we can say what color the light should be. 434 nm of light is the same as 4.34 x 10^-7 meters and according to the chart is BLUE light.
c. When electrons move from n=6 to n=3, energy is also released. However, it is not released in the form of visible light, but INFRARED light.
Using your Reference Table, you should be able to explain what happens when electrons are grounded (releasing energy) or excited by absorbing energy. When electrons are pulled closer to the nucleus, energy is emitted (sometimes in the form of visible light, and at other times in the form of invisible energy (infrared radiation, ultraviolet radiation). When electrons absorb energy, they move away from the nucleus of the atom. Any energy that is emitted from electrons is made up of particles called PHOTONS.
a. For example, when electrons move from n = 2 to n =6, the electrons become more excited because they have ABSORBED energy.
b. When electrons move from n = 5 to n = 2, electrons are GROUNDED and energy is RELEASED. Specifically, 434 nanometers of light are emitted, which is visible light. Because it is visible, we can say what color the light should be. 434 nm of light is the same as 4.34 x 10^-7 meters and according to the chart is BLUE light.
c. When electrons move from n=6 to n=3, energy is also released. However, it is not released in the form of visible light, but INFRARED light.
Objective 1.1.4: Explain the process of radioactive decay using nuclear equations and half-life.
Types of Nuclear Equations:
- Fusion vs. Fission:
Test #2 Content
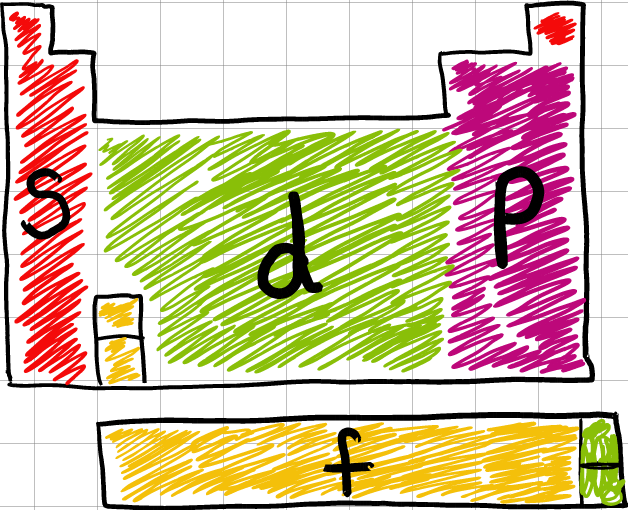
Objective 1.3.1: Classify the components of a periodic table (period, group, metal, metalloid, nonmetal, transition).
Objective 1.3.2: Infer the physical properties (atomic radius, metallic, and nonmetallic characteristics) of an element based on its position on the Periodic Table.
Remember, there are more METALS on the periodic table than any other kind of element. If you locate the bold, stair-step zigzag on the right side of the periodic table, the metals would found to the left of that zigzag. NONMETALS are found to the right of that zigzag (with one exception: Hydrogen). The metalloids are found along the steps of that zigzag.
Objective 1.3.2: Infer the physical properties (atomic radius, metallic, and nonmetallic characteristics) of an element based on its position on the Periodic Table.
Remember, there are more METALS on the periodic table than any other kind of element. If you locate the bold, stair-step zigzag on the right side of the periodic table, the metals would found to the left of that zigzag. NONMETALS are found to the right of that zigzag (with one exception: Hydrogen). The metalloids are found along the steps of that zigzag.
|
Characteristics of Metals:
*They are good CONDUCTORS (transfer energy). *They are typically SOLID (with the exception of Hg) at room temp. *The are very hard, allowing them to be MALLEABLE (can be hammered into thin sheets) and DUCTILE (can be spun into wire) *They form positively charged ions (cations). *They give away electrons. *Some of them (Fe, Co, Ni) are magnetic, so they can attract other metals. |
Characteristics of Nonmetals:
*They are good INSULATORS (stop the transfer of energy). *They can be solid, liquid or gas at room temp, but most are gas. *They are very brittle--if you try to hammer them or spin them into wire, they'll just break if they are solid; liquids and gases would be impossible to do this to. *They form negatively charged ions (anions). *They take electrons. |
Characteristics of Metalloids
*They are SEMICONDUCTORS (some of them can transfer small amounts of energy).
*Some of them are slightly malleable.
*Sometimes they give electrons, and sometimes they take electrons.
*They are SEMICONDUCTORS (some of them can transfer small amounts of energy).
*Some of them are slightly malleable.
*Sometimes they give electrons, and sometimes they take electrons.
The periodic table is divided into GROUPS and PERIODS.
*GROUPS (aka families) are the columns of the table. Elements in the same group have the same number of VALENCE electrons. These elements have similar chemical properties.
*PERIODS are the rows of the table. Elements in the same period demonstrate trends (or changes in characteristics across the row). All elements in the same period have the same number of energy levels (rings).
*GROUPS (aka families) are the columns of the table. Elements in the same group have the same number of VALENCE electrons. These elements have similar chemical properties.
*PERIODS are the rows of the table. Elements in the same period demonstrate trends (or changes in characteristics across the row). All elements in the same period have the same number of energy levels (rings).
Objective 1.3.3: Infer the atomic size, reactivity, electronegativity, and ionization energy of an element from its position on the Periodic Table.
Trends to know:
*Atomic size (the diameter of the atom) DECREASES from LEFT TO RIGHT, and INCREASES from TOP TO BOTTOM.
Why? As we most across a period, elements pick up more electrons which create a greater attraction to the nucleus (think a very strong hug) and that results in a tighter hold and in effect is like the atom "sucking in its gut". Atomic size grows as you move down groups because you pick up a new ring/energy level with each period.
*Electronegativity (the likelihood of an atom to attract an electron) INCREASES from LEFT TO RIGHT and DECREASES from TOP TO BOTTOM.
Why? Metals are positive, and have no desire for more electrons--in fact, they want to get rid of the extras they have on their valence ring. Nonmetals would love more electrons and attract them, so the right side of the PT is much more electronegative than the left. However, the NOBLE GASES are not interested in giving or taking electrons because they are already stable. Therefore, they have 0 electronegativity.
*Ionization Energy (the energy needed to lose an electron) INCREASES from LEFT TO RIGHT and DECREASES from TOP TO BOTTOM.
Why? Metals want to give up their electrons, so it takes relatively little energy for them to do so. Nonmetals aren't trying to lose any electrons, they want more...so it would require lots of energy in order to take one of theirs.
Trends to know:
*Atomic size (the diameter of the atom) DECREASES from LEFT TO RIGHT, and INCREASES from TOP TO BOTTOM.
Why? As we most across a period, elements pick up more electrons which create a greater attraction to the nucleus (think a very strong hug) and that results in a tighter hold and in effect is like the atom "sucking in its gut". Atomic size grows as you move down groups because you pick up a new ring/energy level with each period.
*Electronegativity (the likelihood of an atom to attract an electron) INCREASES from LEFT TO RIGHT and DECREASES from TOP TO BOTTOM.
Why? Metals are positive, and have no desire for more electrons--in fact, they want to get rid of the extras they have on their valence ring. Nonmetals would love more electrons and attract them, so the right side of the PT is much more electronegative than the left. However, the NOBLE GASES are not interested in giving or taking electrons because they are already stable. Therefore, they have 0 electronegativity.
*Ionization Energy (the energy needed to lose an electron) INCREASES from LEFT TO RIGHT and DECREASES from TOP TO BOTTOM.
Why? Metals want to give up their electrons, so it takes relatively little energy for them to do so. Nonmetals aren't trying to lose any electrons, they want more...so it would require lots of energy in order to take one of theirs.
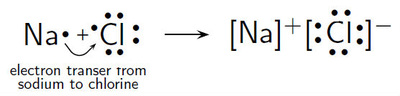
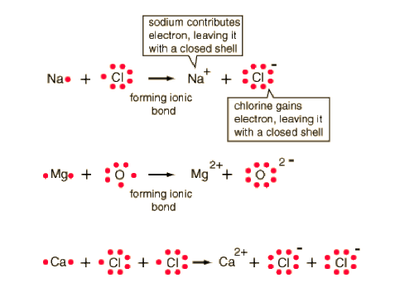
Objective 1.2.1: Compare (qualitatively) the relative strengths of ionic, covalent, and metallic bonds.
*IONIC Bonds: occur when electrons are lost from CATIONs and gained by ANIONs.
*COVALENT Bonds: occur when electrons are SHARED.
*METALLIC Bonds: don't make new compounds...electrons are just free to swirl around positively charged metal ions. (like a sea of electrons!)
Elements are STABLE, according to the octet rule, when they have lost all valence electrons OR gain enough valence electrons to have 8, like the configuration of Noble Gases.
The LEWIS diagrams below show the formation of IONIC and COVALENT bonds in compounds and diatomic molecules. (Click on them to see them clearly).
*IONIC Bonds: occur when electrons are lost from CATIONs and gained by ANIONs.
*COVALENT Bonds: occur when electrons are SHARED.
*METALLIC Bonds: don't make new compounds...electrons are just free to swirl around positively charged metal ions. (like a sea of electrons!)
Elements are STABLE, according to the octet rule, when they have lost all valence electrons OR gain enough valence electrons to have 8, like the configuration of Noble Gases.
The LEWIS diagrams below show the formation of IONIC and COVALENT bonds in compounds and diatomic molecules. (Click on them to see them clearly).
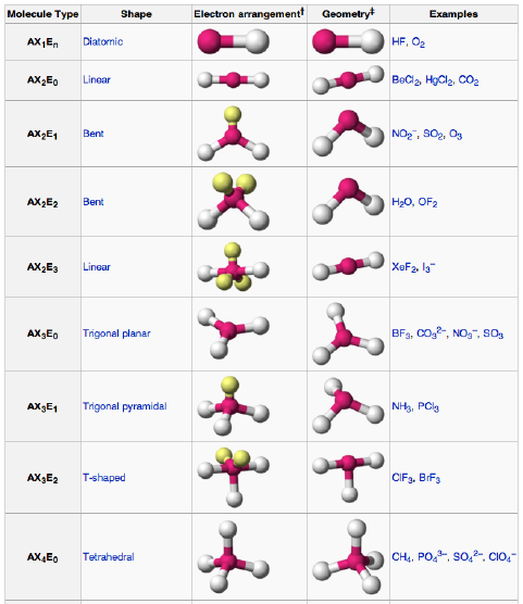
Objective 1.2.2: Infer the type of bond and chemical formula formed between atoms.
*IONIC bonds form between METALS and NONMETALS (or positively-charged ions and negatively-charged ions).
*COVALENT bonds form between NONMETALS only! (No charges involved).
*METALLIC bonds only occur between individual metals or mixtures of metals (Alloys). There is no chemical bond between them, but they will allow electrons to flow easily. They are a SEA of ELECTRONS surrounding a positively charged nucleus.
The form below gives you a chance to try telling the difference between ionic and covalent compounds.
*IONIC bonds form between METALS and NONMETALS (or positively-charged ions and negatively-charged ions).
*COVALENT bonds form between NONMETALS only! (No charges involved).
*METALLIC bonds only occur between individual metals or mixtures of metals (Alloys). There is no chemical bond between them, but they will allow electrons to flow easily. They are a SEA of ELECTRONS surrounding a positively charged nucleus.
The form below gives you a chance to try telling the difference between ionic and covalent compounds.
| |||||||
HERE IS YOUR WORK FOR TUES-WED, FEB 21-22!
Tuesday:
| ionic_and_covalent.docx | |
| File Size: | 15 kb |
| File Type: | docx |
Wednesday:
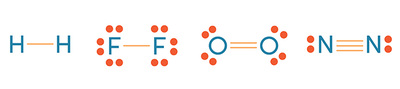
Objective 1.2.3: Compare intra- and inter-particle forces.
Weaker bonds exist that have some effect on molecule's shapes.
*Dipole-dipole forces: slight attractions between polar molecules (the negative end of one and the positive end of another)
*Hydrogen bonds: strong dipole force between Hydrogen and other more electronegative elements (like O, F, N).
*(London) Dispersion forces (also known as van der Waals forces): slight, temporary attraction that results from moving electrons attracted to the positive nucleus of other atoms (kind of like static electricity).
In covalent bonds, three types exist: single, double and triple.
*As the covalent bond # increases, the size shrinks and the strength increases.
*Single bonds are LONG and weak.
*Double bonds are SHORT and strong.
*Triple bonds are VERY SHORT and VERY STRONG (and rare).
Bonds and their Strengths (Weakest to Strongest):
Dispersion < Dipole < Hydrogen < Single Covalent < Ionic < Double Covalent < Triple Covalent
Weaker bonds exist that have some effect on molecule's shapes.
*Dipole-dipole forces: slight attractions between polar molecules (the negative end of one and the positive end of another)
*Hydrogen bonds: strong dipole force between Hydrogen and other more electronegative elements (like O, F, N).
*(London) Dispersion forces (also known as van der Waals forces): slight, temporary attraction that results from moving electrons attracted to the positive nucleus of other atoms (kind of like static electricity).
In covalent bonds, three types exist: single, double and triple.
*As the covalent bond # increases, the size shrinks and the strength increases.
*Single bonds are LONG and weak.
*Double bonds are SHORT and strong.
*Triple bonds are VERY SHORT and VERY STRONG (and rare).
Bonds and their Strengths (Weakest to Strongest):
Dispersion < Dipole < Hydrogen < Single Covalent < Ionic < Double Covalent < Triple Covalent
Objective 1.2.4: Interpret the names and formulas of compounds using IUPAC convention.
Rules for Naming IONIC COMPOUNDS:
1. The cation's name remains the same.
1b. If the cation is a TRANSITION METAL, you must know the charge of the ion, and write it as a roman numeral in the name.
2. The anion's name loses it's ending, and changes to -ide.
3. Any polyatomic ion keeps its name.
4. The number of atoms in the formula doesn't affect the name.
Rules for Naming COVALENT COMPOUNDS:
1. Count the number of each atom in the formula.
2. Use number prefixes in front of the element names to show how many of each element was in the formula.
3. If there is only one atom in the first element in the formula, no prefix is used.
4. Last element in formula will have ending removed and replaced with -ide.
Rules for Naming ACIDS:
1. If acid is made up of only Hydrogen, and one other element: hydro + (root) + ic acid
ex) HF hydrofluoric acid (hydrogen + fluorine)
2. If acid is made up of Hydrogen and an "-ate" polyatomic ion: root + ic acid
ex) H2SO4 sulfuric acid (hydrogen + sulfate)
3. If acid is made up of Hydrogen and an "ite" polyatomic ion: root + ous acid
ex) H2SO3 sulfurous acid (hydrogen + sulfite)
Rules for Naming IONIC COMPOUNDS:
1. The cation's name remains the same.
1b. If the cation is a TRANSITION METAL, you must know the charge of the ion, and write it as a roman numeral in the name.
2. The anion's name loses it's ending, and changes to -ide.
3. Any polyatomic ion keeps its name.
4. The number of atoms in the formula doesn't affect the name.
Rules for Naming COVALENT COMPOUNDS:
1. Count the number of each atom in the formula.
2. Use number prefixes in front of the element names to show how many of each element was in the formula.
3. If there is only one atom in the first element in the formula, no prefix is used.
4. Last element in formula will have ending removed and replaced with -ide.
Rules for Naming ACIDS:
1. If acid is made up of only Hydrogen, and one other element: hydro + (root) + ic acid
ex) HF hydrofluoric acid (hydrogen + fluorine)
2. If acid is made up of Hydrogen and an "-ate" polyatomic ion: root + ic acid
ex) H2SO4 sulfuric acid (hydrogen + sulfate)
3. If acid is made up of Hydrogen and an "ite" polyatomic ion: root + ous acid
ex) H2SO3 sulfurous acid (hydrogen + sulfite)
Objective 1.2.5: Compare the properties of ionic, covalent, metallic, and network compounds.
|
Polar v. Nonpolar
Remember, Polar molecules demonstrate an uneven sharing of electrons, while nonpolar molecules share equally. Make sure you determine whether there are slight negative or slight positive charges on the ends of each OUTER atom in the molecules before making your decision. |
Test #3 Content
Objective 2.2.2: Analyze the evidence of a chemical change.
You should be able to tell the difference between physical and chemical changes. During a physical change, the atomic structure of the substances remains intact. During chemical changes, some new arrangement of the atoms occurs.
You should be able to tell the difference between physical and chemical changes. During a physical change, the atomic structure of the substances remains intact. During chemical changes, some new arrangement of the atoms occurs.
Physical Changes include:
*Changes in size or shape
*Changes in phase/state (solid, liquid, gas)
*Changes in color (ONLY IF A DYE IS USED or a chemical is diluted)
*Changes in size or shape
*Changes in phase/state (solid, liquid, gas)
*Changes in color (ONLY IF A DYE IS USED or a chemical is diluted)
Chemical Changes include:
*Sudden changes in temperature, when you didn't physically alter the temperature
*Changes in color (when no colored chemicals were used)
*Release of a gas (like oxygen or carbon dioxide)
*Formation of a precipitate (a solid) where no solid out of the mixture of two solutions (liquids)
*Release (or absorption) of energy (light, sound, heat)
*Sudden changes in temperature, when you didn't physically alter the temperature
*Changes in color (when no colored chemicals were used)
*Release of a gas (like oxygen or carbon dioxide)
*Formation of a precipitate (a solid) where no solid out of the mixture of two solutions (liquids)
*Release (or absorption) of energy (light, sound, heat)
Objective 2.2.4: Analyze the stoichiometric relationships inherent in a chemical reaction.
|
A mole is a unit of substance.
To find the molar mass (aka molar weight) of a substance, you need to use the periodic table to find the average atomic mass of each part of the substance and multiple each mass by the number of atoms of substance present. Then, you add all of your individual elements' masses together. Ex) NaCl-- Na 22.99, Cl 35.45 22.99 + 35.45 = 58.44 grams/mole Ex 2) Na2SO4 -- Na 22.99 x 2, S 32.07, O 16.00 x 4 45.98 + 32.07 + 64.00 = 142.05 grams/mole Ex 3) Mg(ClO4)2 -- Mg 24.31, Cl 35.45 x 2, O 16.00 x 4 x 2 24.31 + 70.90 + 96.00 = 191.21 grams/mole Click HERE for more practice problems involving Calculating Molar Mass. |
Once you know the molar mass of a substance, if you are asked to find the amount of substance given varying numbers of moles, you can do that simply by multiplying the molar mass by the number of moles you've been given.
|
Once you know the Molar Mass of a substance, you can use it to calculate how many moles of substance are in a given number of grams. You could also use MM to calculate how many grams of a substance are in a given number of moles.
MOLE CONVERSIONS:
One mole of an element is equal to 6.02 x 10^23 atoms of that element.
One mole of a compound substance is equal to 6.02 x 10^23 molecules of that substance.
Avogadro's Number is a constant used in stoichiometry. It simply means that in every unit of substance, there are 6.02 x 10^23 subunits of that substance.
When working with GASES, one mole of the gas takes up 22.4 liters of space at STP (Standard Temperature and Pressure). NOTE: THIS ONLY WORKS FOR GASES AT STP!
One mole of an element is equal to 6.02 x 10^23 atoms of that element.
One mole of a compound substance is equal to 6.02 x 10^23 molecules of that substance.
Avogadro's Number is a constant used in stoichiometry. It simply means that in every unit of substance, there are 6.02 x 10^23 subunits of that substance.
When working with GASES, one mole of the gas takes up 22.4 liters of space at STP (Standard Temperature and Pressure). NOTE: THIS ONLY WORKS FOR GASES AT STP!
Here are some practice problems that you can try if you wish to prepare for Mole Conversions.
1. How many moles are in 32.5 grams of NaO?
2. How many molecules are in 9.33 grams of K2S?
3. How many atoms of Nitrogen are in 16.2 moles of N2?
4. How many grams are in 2.55 x 10^24 molecules of Mg(OH)2?
5. How many liters are in 8.22 x 10^23 molecules of N2?
1. How many moles are in 32.5 grams of NaO?
2. How many molecules are in 9.33 grams of K2S?
3. How many atoms of Nitrogen are in 16.2 moles of N2?
4. How many grams are in 2.55 x 10^24 molecules of Mg(OH)2?
5. How many liters are in 8.22 x 10^23 molecules of N2?
Practice: Mole Problems!
Objective 2.2.5: Analyze quantitatively the composition of a substance (empirical formula, molecular formula, percent composition, and hydrates).
|
Percent Composition (by mass) is simply used to tell what % of each element makes up a compound based on the mass of that element within the compound. In order to complete Percent Composition problems, you simply have to divide the mass of each element involved in the substance (and how many times that element occurs) by the Molar Mass of the whole compound. Then, you multiply by 100 to get the percent.
For example, Ca(OH)2: 1 Ca = 40.08 2 O = 16.00 x 2 = 32.00 2 H = 1.008 x 2 = 2.016 Molar Mass = 74.096 Ca % = 40.08/74.096 = 0.5409 x 100 = 54.09% O% = 32.00/74.096 = 0.4319 x 100 = 43.19% H% = 2.016/74.096 = 0.0272 x 100 = 2.72% *Note: The % should add up to be very close to 100%! |
To calculate the identity of an unknown hydrate:
1. Calculate the molar masses of the hydrates available.
2. Calculate the % water of those hydrates.
3. Subtract the mass of the empty crucible from the masses of a) the crucible with unknown hydrate BEFORE flaming and b) the crucible with the unknown hydrate AFTER flaming. Subtract b from a to determine how much water was lost from the unknown. Then, divide that answer by the BEFORE result. Multiply by 100. This should give you the % of water that was lost from the flaming process.
4. Compare that percentage to the percentages of water in the available hydrates.
5. Choose the % closest to your result.
6. Perform a Percent Error calculation to see how close your result was. Remember, Percent Error is (Experimental - Expected)/Expected, then multiply by 100. Values less than 1% have high confidence. Values less than 5% have moderate confidence. Values less than 10% have low confidence. Values above 10% have minimal confidence.
Example:
Three hydrates were presented to a student: NaC2H3O2*3H2O, CaSO4*2H2O, and Zn(NO3)2*6H2O.
(Molar masses, respectively: 136.08, 172.17, 297.48)
% Water in the Hydrates, respectively: 54.05/136.08x100= 39.72%, 36.03/172.17x100= 20.93%, 108.10/297.48x100= 36.34%
An empty crucible was massed to be 12.7 grams.
The crucible with unknown before flaming had a mass of: 36.8 grams.
The crucible with unknown after flaming had a mass of 30.7 grams.
36.8-12.7 = 24.1
30.7-12.7 = 18.0
24.1-18.0 = 6.1
6.1/24.1x100 = 25.31%
The unknown (25.31%) is closest to 20.93%, so the hydrate must be Calcium Sulfate Dihydrate (CaSO4*2H2O).
Percent Error = 25.31 - 20.93 / 20.93 x 100 = 4.38/20.93 x 100 = 0.2093 x 100 = 20.93%
There is little confidence in the result, but it is the most likely result out of the three.
1. Calculate the molar masses of the hydrates available.
2. Calculate the % water of those hydrates.
3. Subtract the mass of the empty crucible from the masses of a) the crucible with unknown hydrate BEFORE flaming and b) the crucible with the unknown hydrate AFTER flaming. Subtract b from a to determine how much water was lost from the unknown. Then, divide that answer by the BEFORE result. Multiply by 100. This should give you the % of water that was lost from the flaming process.
4. Compare that percentage to the percentages of water in the available hydrates.
5. Choose the % closest to your result.
6. Perform a Percent Error calculation to see how close your result was. Remember, Percent Error is (Experimental - Expected)/Expected, then multiply by 100. Values less than 1% have high confidence. Values less than 5% have moderate confidence. Values less than 10% have low confidence. Values above 10% have minimal confidence.
Example:
Three hydrates were presented to a student: NaC2H3O2*3H2O, CaSO4*2H2O, and Zn(NO3)2*6H2O.
(Molar masses, respectively: 136.08, 172.17, 297.48)
% Water in the Hydrates, respectively: 54.05/136.08x100= 39.72%, 36.03/172.17x100= 20.93%, 108.10/297.48x100= 36.34%
An empty crucible was massed to be 12.7 grams.
The crucible with unknown before flaming had a mass of: 36.8 grams.
The crucible with unknown after flaming had a mass of 30.7 grams.
36.8-12.7 = 24.1
30.7-12.7 = 18.0
24.1-18.0 = 6.1
6.1/24.1x100 = 25.31%
The unknown (25.31%) is closest to 20.93%, so the hydrate must be Calcium Sulfate Dihydrate (CaSO4*2H2O).
Percent Error = 25.31 - 20.93 / 20.93 x 100 = 4.38/20.93 x 100 = 0.2093 x 100 = 20.93%
There is little confidence in the result, but it is the most likely result out of the three.
Empirical and Molecular Formulas
The empirical formula of a compound is the SIMPLEST ratio of the compound. The molecular formula can sometimes be the same as the empirical formula, but often is a nonreduced formula consisting of more atoms because they are needed to complete the actual structure of the compound.
To Calculate Empirical Formula for a compound:
1. Change percentages into grams (No conversion, just change the sign.)
2. Turn the grams of each element into moles.
3. Identify the element with the lowest number of moles and divide all of the elements' mole value by the lowest.
4. Look for "pretty numbers" (whole numbers, simple fractions [1/2, 1/3, etc...]).
5. If the "pretty numbers" are all whole numbers, those are the number of atoms of each element in the compound.
5b. If the "pretty numbers" include fractions, you must multiply the pretty numbers of all of the elements by the denominator of the fraction. Now, you should have all whole numbers and those are the numbers of atoms of each element of the compound.
To Calculate Molecular Formula for a compound:
1. Follow the same steps for empirical formula.
2. Calculate the molar mass of the empirical formula.
3. You should be given another molar mass of the molecular formula. Divide the molar mass of the molecular formula by the molar mass of the empirical formula. If it equals one, then the empirical and molecular formulas are the same. If it equals any other whole number value, use that value and multiply each of the elements and their numbers of atoms by that value.
The empirical formula of a compound is the SIMPLEST ratio of the compound. The molecular formula can sometimes be the same as the empirical formula, but often is a nonreduced formula consisting of more atoms because they are needed to complete the actual structure of the compound.
To Calculate Empirical Formula for a compound:
1. Change percentages into grams (No conversion, just change the sign.)
2. Turn the grams of each element into moles.
3. Identify the element with the lowest number of moles and divide all of the elements' mole value by the lowest.
4. Look for "pretty numbers" (whole numbers, simple fractions [1/2, 1/3, etc...]).
5. If the "pretty numbers" are all whole numbers, those are the number of atoms of each element in the compound.
5b. If the "pretty numbers" include fractions, you must multiply the pretty numbers of all of the elements by the denominator of the fraction. Now, you should have all whole numbers and those are the numbers of atoms of each element of the compound.
To Calculate Molecular Formula for a compound:
1. Follow the same steps for empirical formula.
2. Calculate the molar mass of the empirical formula.
3. You should be given another molar mass of the molecular formula. Divide the molar mass of the molecular formula by the molar mass of the empirical formula. If it equals one, then the empirical and molecular formulas are the same. If it equals any other whole number value, use that value and multiply each of the elements and their numbers of atoms by that value.
Objective 2.2.3: Analyze the law of conservation of matter and how it applies to various types of chemical equations (aka Know how to classify and balance equations).
Types of Equations:
1. Synthesis: These equations show multiple elements/compounds being added together to make one or more products. However, there are always more reactants than products.
Ex. 1) 2Mg + O2 --> 2MgO
Ex. 2) 2H2 + O2 --> 2H2O
2. Decomposition: These equations show a compound being broken down into multiple products. There are always more products than reactants.
Ex. 1) 2H2O2 --> 2H2O + O2
Ex. 2) 4KClO3 --> 3KClO4 + KCl
3. Single Replacement: These equations show a compound added to a single element. The element replaces its equivalent within the compound and frees the complimentary element. However, you must consult the ACTIVITY SERIES chart within your yellow reference table. The single element can only replace the complimentary element in the compound if it is higher on the ACTIVITY SERIES chart.
Ex. 1) 2NaCl + F2 --> 2NaF + Cl2 (Notice: the F replaces the Cl because it's higher on the ACTIVITY SERIES chart.)
Ex. 2) MgO + 2Li --> Li2O + Mg (Notice: the Li replaces the Mg because it's higher on the ACTIVITY SERIES chart.)
4. Double Replacement: These equations show two compounds being added together and the partners swap to produce two new products.
Ex. 1) MgCl2 + Na2O --> MgO + 2NaCl
Ex. 2) HCl + NaOH --> NaCl + HOH
5. Combustion: These equations show what happens whenever a hydrocarbon fuel is ignited in the presence of oxygen gas. It ALWAYS produces carbon dioxide and water vapor as products.
Ex. 1) C3H8 + 5O2 --> 3CO2 +4 H2O
Ex. 2) 2C4H10 + 13O2 --> 8CO2 +10H2O
1. Synthesis: These equations show multiple elements/compounds being added together to make one or more products. However, there are always more reactants than products.
Ex. 1) 2Mg + O2 --> 2MgO
Ex. 2) 2H2 + O2 --> 2H2O
2. Decomposition: These equations show a compound being broken down into multiple products. There are always more products than reactants.
Ex. 1) 2H2O2 --> 2H2O + O2
Ex. 2) 4KClO3 --> 3KClO4 + KCl
3. Single Replacement: These equations show a compound added to a single element. The element replaces its equivalent within the compound and frees the complimentary element. However, you must consult the ACTIVITY SERIES chart within your yellow reference table. The single element can only replace the complimentary element in the compound if it is higher on the ACTIVITY SERIES chart.
Ex. 1) 2NaCl + F2 --> 2NaF + Cl2 (Notice: the F replaces the Cl because it's higher on the ACTIVITY SERIES chart.)
Ex. 2) MgO + 2Li --> Li2O + Mg (Notice: the Li replaces the Mg because it's higher on the ACTIVITY SERIES chart.)
4. Double Replacement: These equations show two compounds being added together and the partners swap to produce two new products.
Ex. 1) MgCl2 + Na2O --> MgO + 2NaCl
Ex. 2) HCl + NaOH --> NaCl + HOH
5. Combustion: These equations show what happens whenever a hydrocarbon fuel is ignited in the presence of oxygen gas. It ALWAYS produces carbon dioxide and water vapor as products.
Ex. 1) C3H8 + 5O2 --> 3CO2 +4 H2O
Ex. 2) 2C4H10 + 13O2 --> 8CO2 +10H2O
Using your Yellow Reference Tables, you can predict the products of equations when you are given only the reactants.Here's some practice in predicting products:
| |||||||
Net Ionic Equations (only used when dealing with Double Replacement Reactions)
We perform Net Ionic equations to see if a solid precipitate is created when a double replacement reaction occurs after mixing two aqueous solutions together.
How to Do Them:
Step 1: Take a Double Replacement Reaction:
NaOH + MgCl2 --> Mg(OH)2 + NaCl
Step 2: Balance the equation.
2NaOH + MgCl2 --> Mg(OH)2 + 2 NaCl
Step 3: Remember, the compounds on the left are aqueous (aq) so they dissolve in a solution and natural break down into the ions that were making them up. However, now you have to look at the Solubility Rules on the Yellow Reference Table to see about your products.
Step 4: Solubility Rules:
2NaOH (aq) + MgCl2 (aq) --> Mg(OH)2 (s) +2 NaCl (aq) *Remember, the (s) means SOLID. This is a precipitate!
Step 6: To make the net ionic equation, we need to break up all of the ions for the aqueous (aq) compounds.
2Na+(aq) + 2OH- (aq) + Mg2+ (aq) + 2Cl- (aq) --> Mg(OH)2 (s) + 2Na+ (aq) + 2Cl- (aq)
Step 7: Mark out all of those ions that are the EXACT SAME on both sides. These are called spectator ions and they do not contribute to the actual reaction.
In this case, 2Na+ (aq) and 2Cl- (aq) are the spectator ions are not needed.
So now, our final net ionic equation is this: Mg2+ (aq) + 2OH- (aq) --> Mg(OH)2 (s)
Sometimes, you will get through Step 3 and see that everything is aqueous (aq). In that case, there is NO NET IONIC equation and you stop there, with the balanced equation.
We perform Net Ionic equations to see if a solid precipitate is created when a double replacement reaction occurs after mixing two aqueous solutions together.
How to Do Them:
Step 1: Take a Double Replacement Reaction:
NaOH + MgCl2 --> Mg(OH)2 + NaCl
Step 2: Balance the equation.
2NaOH + MgCl2 --> Mg(OH)2 + 2 NaCl
Step 3: Remember, the compounds on the left are aqueous (aq) so they dissolve in a solution and natural break down into the ions that were making them up. However, now you have to look at the Solubility Rules on the Yellow Reference Table to see about your products.
Step 4: Solubility Rules:
- Mg(OH)2: All hydroxides except Group 1A, Strontium and Ammonium are INSOLUBLE. This means they WILL make a solid and WILL NOT DISSOLVE in the solution.
- NaCl: All chlorides are SOLUBLE. This means that NaCl will dissolve to make an aqueous solution.
2NaOH (aq) + MgCl2 (aq) --> Mg(OH)2 (s) +2 NaCl (aq) *Remember, the (s) means SOLID. This is a precipitate!
Step 6: To make the net ionic equation, we need to break up all of the ions for the aqueous (aq) compounds.
2Na+(aq) + 2OH- (aq) + Mg2+ (aq) + 2Cl- (aq) --> Mg(OH)2 (s) + 2Na+ (aq) + 2Cl- (aq)
Step 7: Mark out all of those ions that are the EXACT SAME on both sides. These are called spectator ions and they do not contribute to the actual reaction.
In this case, 2Na+ (aq) and 2Cl- (aq) are the spectator ions are not needed.
So now, our final net ionic equation is this: Mg2+ (aq) + 2OH- (aq) --> Mg(OH)2 (s)
Sometimes, you will get through Step 3 and see that everything is aqueous (aq). In that case, there is NO NET IONIC equation and you stop there, with the balanced equation.
Objective 2.2.4: Analyze the stoichiometric relationships inherent in a chemical reaction.
Remember, in this section, we just put two of our favorite things together: balancing equations and the fences! We can use stoichiometry to make calculations about different substances within the equation after applying the mole-mole ratio.
Ex) How many grams of oxygen gas are needed to burn 12.5 L of octane (C8H18)?
FIRST, we must figure out what kind of equation we are building and what the general equation will look like. Look for given substances and key terms in the problem.
How many grams of oxygen gas are needed to burn 12.5L of octane (C8H18)?
12.5 L C8H18 x 1 mole C8H18 x 25 moles O2 x 32.0 grams O2 = 223 grams O2 (3 sig figs)
22.4 L C8H18 2 moles C8H18 1 mole O2
Ex 2) How many molecules of water will be created after 35 grams of HCl are combined with Mg(OH)2 ?
Step 1:
35 g HCl x 1 mole HCl x 1 mole H2O x 6.02 x 10E23 m/c H2O = 2.9 x 10E23 (2 sig figs)
36.458 grams HCl 2 moles HCl 1 mole H2O
Ex) How many grams of oxygen gas are needed to burn 12.5 L of octane (C8H18)?
FIRST, we must figure out what kind of equation we are building and what the general equation will look like. Look for given substances and key terms in the problem.
How many grams of oxygen gas are needed to burn 12.5L of octane (C8H18)?
- Substances we know are in the equation: O2, C8H18
- Action happening: burning
- Going from 12.5 L of C8H18 to ?g of O2.
- Since you have a hydrocarbon fuel, and oxygen, and it's burning...it must be COMBUSTION!
- The equation then gets set up as a combustion reaction: C8H18 + O2 --> CO2 + H2O
- Even though we weren't given the products, we know carbon dioxide and water are always the products of a combustion reaction.
- 2 C8H8 + 25 O2 --> 16 CO2 + 18 H2O
12.5 L C8H18 x 1 mole C8H18 x 25 moles O2 x 32.0 grams O2 = 223 grams O2 (3 sig figs)
22.4 L C8H18 2 moles C8H18 1 mole O2
Ex 2) How many molecules of water will be created after 35 grams of HCl are combined with Mg(OH)2 ?
Step 1:
- Substances in the reaction: water H2O, HCl, Mg(OH)2
- Action: combined
- Going from 35 grams HCl to ? molecules H2O
- HCl and Mg(OH)2 are combined to make H2O
- We see Cl and Mg haven't been mentioned again, so we know they must combine too (MgCl2)
- 2 compounds combining to make 2 more compounds must be a double replacement!
- The equation must be: HCl + Mg(OH)2 --> MgCl2 + H2O
- 2HCl + Mg(OH)2 --> MgCl2 + H2O (after balancing)
35 g HCl x 1 mole HCl x 1 mole H2O x 6.02 x 10E23 m/c H2O = 2.9 x 10E23 (2 sig figs)
36.458 grams HCl 2 moles HCl 1 mole H2O
Practice Problems:
See if you can come up with the answers located at the end of these practice problems!
1) How many moles of F2 gas must react with AlCl3 in order to produce 25.1 L of Cl2 gas?
2) How many molecules of CO2 will be produced when 55 grams of C3H8 is burned?
3) How many grams of HgO will be made when 48 grams of Hg are used?
4) How many atoms of Zn are freed when Lithium is added to 20.0 grams of zinc chloride?
5) How many liters of H2 gas are released when 5 moles of HCl are decomposed?
Answers (in correct sig figs):
1) 1.12 moles F2
2) 2.3E24 m/c CO2
3) 52 g HgO
4) 8.83E22 atoms Zn
5) 60 liters H2
See if you can come up with the answers located at the end of these practice problems!
1) How many moles of F2 gas must react with AlCl3 in order to produce 25.1 L of Cl2 gas?
2) How many molecules of CO2 will be produced when 55 grams of C3H8 is burned?
3) How many grams of HgO will be made when 48 grams of Hg are used?
4) How many atoms of Zn are freed when Lithium is added to 20.0 grams of zinc chloride?
5) How many liters of H2 gas are released when 5 moles of HCl are decomposed?
Answers (in correct sig figs):
1) 1.12 moles F2
2) 2.3E24 m/c CO2
3) 52 g HgO
4) 8.83E22 atoms Zn
5) 60 liters H2
More Stoichiometry:
Using stoichiometry, you can move between various units such as: liters of gas, atoms, molecules, grams, and moles. Remember, if you have trouble, just use the map to guide your way through the problems. Examples of problems can be accessed using the button below. The solutions are found on the button to its right.
Using stoichiometry, you can move between various units such as: liters of gas, atoms, molecules, grams, and moles. Remember, if you have trouble, just use the map to guide your way through the problems. Examples of problems can be accessed using the button below. The solutions are found on the button to its right.
Objective 2.2.1: Explain the energy content of a chemical reaction.
Reactions happen because molecules collide with one another with enough energy to break bonds, rearrange atoms and reform new bonds. This idea is called collision theory.
Factors that affect collision theory:
1. Temperature: Higher temperatures cause faster molecular movement, and more collisions. Lower temperatures slow down particle movement and reduce collisions.
2. Pressure: Higher pressures cause GASES to move faster resulting in more collisions. Lower pressures allow GASES to slow down and result in less collisions.
Factors that affect collision theory:
1. Temperature: Higher temperatures cause faster molecular movement, and more collisions. Lower temperatures slow down particle movement and reduce collisions.
2. Pressure: Higher pressures cause GASES to move faster resulting in more collisions. Lower pressures allow GASES to slow down and result in less collisions.
- With solids and liquids, pressure has no effect. However, then having a higher concentration will cause more collisions and a lower concentration will reduce collisions.
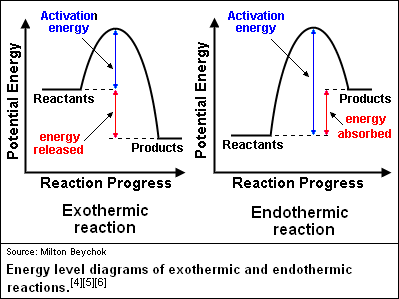
Endothermic v. Exothermic Reactions
Exothermic reactions occur when energy is released during the reaction. Because of this, these reactions feel warm or hot because heat is being released. Notice the products have less energy than the reactants, because much of the energy was released.
Endothermic reactions occur when energy is absorbed during the reaction. Because of this, these reactions feel cold. Notice the products have more energy than the reactants because energy was absorbed.
The activation energy is the amount of energy that must be present for the reaction to occur. Notice that endothermic reactions require more activation energy than exothermic reactions. Special chemicals called CATALYSTS can reduce the activation energy and allow the reaction to occur with less energy. (NOTE: catalysts DO NOT add energy to the reaction!)
Heat v. Temperature
Remember, HEAT and TEMPERATURE are NOT the same! Heat is a type of ENERGY. Temperature is a measurement of the AVERAGE KINETIC ENERGY of the particles of a substance. Think of something that you perceive as cold, such as ice. Even though ice is cold, it absorbs heat energy which breaks hydrogen bonds because the particles speed up allowing them to break bonds and become a liquid.
Exothermic reactions occur when energy is released during the reaction. Because of this, these reactions feel warm or hot because heat is being released. Notice the products have less energy than the reactants, because much of the energy was released.
Endothermic reactions occur when energy is absorbed during the reaction. Because of this, these reactions feel cold. Notice the products have more energy than the reactants because energy was absorbed.
The activation energy is the amount of energy that must be present for the reaction to occur. Notice that endothermic reactions require more activation energy than exothermic reactions. Special chemicals called CATALYSTS can reduce the activation energy and allow the reaction to occur with less energy. (NOTE: catalysts DO NOT add energy to the reaction!)
Heat v. Temperature
Remember, HEAT and TEMPERATURE are NOT the same! Heat is a type of ENERGY. Temperature is a measurement of the AVERAGE KINETIC ENERGY of the particles of a substance. Think of something that you perceive as cold, such as ice. Even though ice is cold, it absorbs heat energy which breaks hydrogen bonds because the particles speed up allowing them to break bonds and become a liquid.
Objective 3.1.1: Explain the factors that affect the rate of a reaction (temperature, concentration, particle size, and presence of a catalyst).
The rate of reaction is all about the SPEED that reactants are converted to products and vice versa--NOT the concentrations of the reactants or products! Reactions occur because collisions between particles happen. Strong collisions will break bonds and separate particles, releasing energy and allowing new bonds to form.
There are several factors that can affect the rate of reactions:
1. Temperature. By increasing the temperature (average kinetic energy of particles), we increase the motion and speed of the moving particles which allow for more collisions.
2. Concentration. By increasing the concentration of particles, we put more particles into a certain area increasing the chances of collisions between those particles.
3. Particle size. By increasing the surface area of the particles, we make the particles more likely to come in contact with other particles, thereby increasing the number of collisions and the reaction rate.
4. Using catalysts. By using a chemical catalyst, the activation energy needed to start the reaction is lowered making it easier for collisions to happen and making the reaction happen faster.
The rate of reaction is all about the SPEED that reactants are converted to products and vice versa--NOT the concentrations of the reactants or products! Reactions occur because collisions between particles happen. Strong collisions will break bonds and separate particles, releasing energy and allowing new bonds to form.
There are several factors that can affect the rate of reactions:
1. Temperature. By increasing the temperature (average kinetic energy of particles), we increase the motion and speed of the moving particles which allow for more collisions.
2. Concentration. By increasing the concentration of particles, we put more particles into a certain area increasing the chances of collisions between those particles.
3. Particle size. By increasing the surface area of the particles, we make the particles more likely to come in contact with other particles, thereby increasing the number of collisions and the reaction rate.
4. Using catalysts. By using a chemical catalyst, the activation energy needed to start the reaction is lowered making it easier for collisions to happen and making the reaction happen faster.
|
Objective 3.1.2: Explain the conditions of a system at equilibrium.
Remember, in reversible reactions, chemical equilibrium is reached when the rates of the forward and backward reactions are equal. Both sides are reacting at the SAME SPEED. They are NOT in equal concentration--those are always changing as the reversible reactions occur. To calculate the equilibrium constant, use the following equation: Keq = ([C]^c x [D]^d) ([A]^a x [B]^b) |
Remember,
*If Keq > 1, then the products are favored. *If Keq < 1, then the reactants are favored. *If Keq = 1, then neither is favored and the reactions are at equilibrium. Also Remember, *Only substances that are aqueous (aq) or gaseous (g). *Solids (s) and pure liquids (l) are given a value of 1. |
Objective 3.1.3: Infer the shift in equilibrium when a stress is applied to a chemical system (Le Chatelier's Principle).
Le Chatelier's Principle says that any stress on a system in equilibrium will result in a shift in the equilibrium.
Stresses that commonly affect systems in equilibrium include:
1. Changes in concentrations of the Reactants/Products.
>Adding more reactants or products will shift the equation in the direction away from the added substances.
>Removing reactants or products will shift the equation in the direction toward the missing items.
2. Changes in energy.
>If energy is required for a reaction and it is removed, the reaction will shift toward the energetic side of the reaction.
>If energy is required for a reaction and it is added, the reaction will shift toward the side of the reaction with no energy.
>Note: Remember, increasing the temperature increases energy and decreasing the temperature decreases energy.
3. Changes in pressure. (THIS ONLY APPLIES TO GASES!)
>Compare the moles of gases on each side of the equation.
>If pressure is increased, the equation will shift in the direction of the lowest number of total moles of gas.
>If pressure is decreased, the equation will shift toward the side with the highest number of total moles.
Le Chatelier's Principle says that any stress on a system in equilibrium will result in a shift in the equilibrium.
Stresses that commonly affect systems in equilibrium include:
1. Changes in concentrations of the Reactants/Products.
>Adding more reactants or products will shift the equation in the direction away from the added substances.
>Removing reactants or products will shift the equation in the direction toward the missing items.
2. Changes in energy.
>If energy is required for a reaction and it is removed, the reaction will shift toward the energetic side of the reaction.
>If energy is required for a reaction and it is added, the reaction will shift toward the side of the reaction with no energy.
>Note: Remember, increasing the temperature increases energy and decreasing the temperature decreases energy.
3. Changes in pressure. (THIS ONLY APPLIES TO GASES!)
>Compare the moles of gases on each side of the equation.
>If pressure is increased, the equation will shift in the direction of the lowest number of total moles of gas.
>If pressure is decreased, the equation will shift toward the side with the highest number of total moles.
Test #4 Material
Objective 2.1.2: Explain heating and cooling curves (heat of fusion, heat of vaporization, heat, melting point, boiling point).
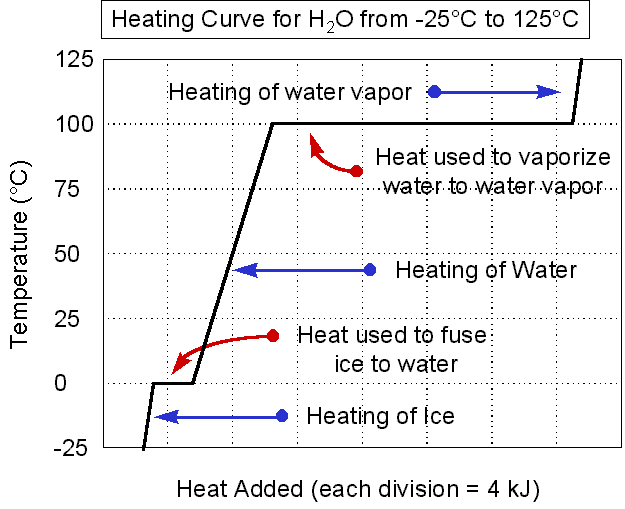
Heating/cooling curves show you the line graph that depicts the transition of a substance from a solid into a liquid, and then into a gas as energy is added (OR vice versa as energy is released).
Heating/cooling curves show you the line graph that depicts the transition of a substance from a solid into a liquid, and then into a gas as energy is added (OR vice versa as energy is released).
|
Look at the Heating Curve on the left.
Notice at temperatures under 0 degrees C, water is a solid. As energy is added, the temperature increases (so molecular movement AKA kinetic energy increases), but the water remains ice until it hits that melting point (0 degrees C). At the melting point, the energy being absorbed no longer raises the temperature because that energy is used to MELT the ice. Once all of the ice is melted, the substance has turned completely to liquid (as seen by the second diagonal line) and temperature begins to rise again as energy is absorbed. Temperature continues to rise until the boiling point (100 degrees C) is reached and once again, molecular motion stops increasing and the energy is used to vaporize the water, turning it into steam. Once all of the water is steam, as energy is absorbed, the temperature begins to rise again. The exact opposite happens as energy is released, and steam will drop in temperature as it approaches the boiling point. Once there, additional energy lost will allow Hydrogen bonds to form again and the gas is converted to a liquid (condensation). Once all gas is liquefied, energy lost results in a falling temperature until the melting point is reached and then energy will be released to the point that the liquid begins freezing. Temperature remains constant during this process, just like it did during melting, and once all of the liquid is frozen, the temperature will resume its dropping nature. |
We can calculate how much heat is absorbed or released using the equations: Q = m(Tf-Ti)Cp, Q =m(Hf), Q = m(Hv).
When phase is NOT changing, we use the long equation: Q = m(Tf-Ti)Cp, thermal energy = mass x change in temp. x specific heat.
For example, suppose we had 125 grams of liquid H2O that warmed from 35.0 degrees to 47.0 degrees. How much thermal energy was ABSORBED for this to happen? (We know it was absorbed because the temp. increased. Therefore, the answer should be positive).
Q = m(Tf - Ti)Cp
Q = 125g (47.0 - 35.0) 4.18* *We know the specific heat of liquid water because it's on our YELLOW REF. TABLE
Q = 6270 Joules
Another example, suppose we had 67.0 grams of ice that we wanted to make even colder. We lowered the temp. from -2.00 degrees C to -5.00 degrees C. How much energy was released from the ice? (We expect a NEGATIVE answer here because energy must be released to lower the temperature).
Q = m(Tf-Ti)Cp
Q = 67.0 (-5.00 - -2.00) 2.05* *We know the specific heat of ice because it's on our YELLOW REF. TABLE
Q = 67.0 (-3.00) 2.05
Q = -412 Joules (Remember, the answer is negative because we released energy.
But what if phases ARE changing? Then, we must use the shorter equations, because if phases are changing, temperature is NOT changing (as seen on the graph above).
What if that 65.0 grams of ice absorbed enough energy to become liquid water? We use the equation Q = mHf, mass x heat of fusion. The heat of fusion is the amount of energy needed to change 1 gram of a solid into a liquid. Every substance has its own unique heat of fusion. Water's heat of fusion is on our YELLOW REF. TABLE.
Q = mHf
Q = 65.0 (334)
Q = 21700 Joules
What if that same amount of water absorbed enough energy to become steam? We use the equation Q = mHv, mass x heat of vaporization. The heat of vaporization is the amount of energy needed to change 1 gram of a liquid into a gas. Every substance has its own unique heat of vaporization. Water's heat of vaporization is on our YELLOW REF. TABLE.
Q = mHv
Q = 65.0 (2260)
Q = 147,000 Joules *Notice MUCH more energy must be absorbed to change a liquid to a gas than a solid to a liquid.
These equations can also be completed if phases are changing because of the release of energy. The only change you would make is that you would make the heat of fusion or heat of vaporization negative.
For example, suppose you had 87.0 grams of liquid water that you wanted to freeze.
Q = m(-Hf)
Q = 87.0 (-334)
Q = -29100 Joules *this value is negative because that's how much energy must be released to freeze the liquid.
When phase is NOT changing, we use the long equation: Q = m(Tf-Ti)Cp, thermal energy = mass x change in temp. x specific heat.
For example, suppose we had 125 grams of liquid H2O that warmed from 35.0 degrees to 47.0 degrees. How much thermal energy was ABSORBED for this to happen? (We know it was absorbed because the temp. increased. Therefore, the answer should be positive).
Q = m(Tf - Ti)Cp
Q = 125g (47.0 - 35.0) 4.18* *We know the specific heat of liquid water because it's on our YELLOW REF. TABLE
Q = 6270 Joules
Another example, suppose we had 67.0 grams of ice that we wanted to make even colder. We lowered the temp. from -2.00 degrees C to -5.00 degrees C. How much energy was released from the ice? (We expect a NEGATIVE answer here because energy must be released to lower the temperature).
Q = m(Tf-Ti)Cp
Q = 67.0 (-5.00 - -2.00) 2.05* *We know the specific heat of ice because it's on our YELLOW REF. TABLE
Q = 67.0 (-3.00) 2.05
Q = -412 Joules (Remember, the answer is negative because we released energy.
But what if phases ARE changing? Then, we must use the shorter equations, because if phases are changing, temperature is NOT changing (as seen on the graph above).
What if that 65.0 grams of ice absorbed enough energy to become liquid water? We use the equation Q = mHf, mass x heat of fusion. The heat of fusion is the amount of energy needed to change 1 gram of a solid into a liquid. Every substance has its own unique heat of fusion. Water's heat of fusion is on our YELLOW REF. TABLE.
Q = mHf
Q = 65.0 (334)
Q = 21700 Joules
What if that same amount of water absorbed enough energy to become steam? We use the equation Q = mHv, mass x heat of vaporization. The heat of vaporization is the amount of energy needed to change 1 gram of a liquid into a gas. Every substance has its own unique heat of vaporization. Water's heat of vaporization is on our YELLOW REF. TABLE.
Q = mHv
Q = 65.0 (2260)
Q = 147,000 Joules *Notice MUCH more energy must be absorbed to change a liquid to a gas than a solid to a liquid.
These equations can also be completed if phases are changing because of the release of energy. The only change you would make is that you would make the heat of fusion or heat of vaporization negative.
For example, suppose you had 87.0 grams of liquid water that you wanted to freeze.
Q = m(-Hf)
Q = 87.0 (-334)
Q = -29100 Joules *this value is negative because that's how much energy must be released to freeze the liquid.
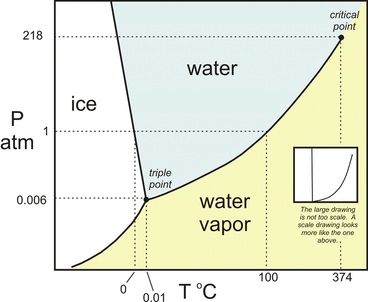
Objective 2.1.3: Interpret the data presented in phase diagrams.
Below, you can see the phase change diagrams for Water (H2O) and Carbon Dioxide (CO2).
Below, you can see the phase change diagrams for Water (H2O) and Carbon Dioxide (CO2).
0.01 degrreeeUsing the phase change diagrams above, you should be able to point out certain things about the substances.
*The first thing you can see is what phase the substance is in at a given temperature and pressure.
For example, what phase is water in at 1 atm and 70 degrees C? It's a liquid! (just look at where those two lines would meet!)
What phase is water in at 1 atm and -10 degrees C? It's a solid!
What about CO2 at 1 atm and 70 degrees C? It's a gas!
What about CO2 at 1 atm and -10 degrees C? Still, a gas!
What about CO2 at 1 atm and -80 degrees C? It's a solid at that very low temperature!
*Using these diagrams you can also describe what happens if one variable is changed but the other is held constant.
For example, what happens to water at 1 atm, if the temperature increases from 25.0 degrees C to 75.0 degrees C? Nothing! It remains liquid! But, what happens if it goes from 25.0 degrees C to 105 degrees C? It changes from liquid to gas.
Also what if you had CO2 at 1 atm and -80 degrees C (it's a gas). However, if the pressure is increased to 5 atm, it changes from gas to solid!
*We can also use these graphs to find the melting and boiling point at a certain pressure.
For example, at 1 atm, the melting point of water is 0 degrees C and the boiling point of water is 100 degrees C.
*Finally, we can use phase change charts to point out the TRIPLE POINT. It's the point where at that given temperature and pressure, it is possible to have ALL 3 PHASES at once! For water, the triple point is at 0.006 atm of pressure, and 0.01 degrees C. For CO2, the triple point is at 5.1 atm of pressure and -56.7 degrees Celsius.
*The first thing you can see is what phase the substance is in at a given temperature and pressure.
For example, what phase is water in at 1 atm and 70 degrees C? It's a liquid! (just look at where those two lines would meet!)
What phase is water in at 1 atm and -10 degrees C? It's a solid!
What about CO2 at 1 atm and 70 degrees C? It's a gas!
What about CO2 at 1 atm and -10 degrees C? Still, a gas!
What about CO2 at 1 atm and -80 degrees C? It's a solid at that very low temperature!
*Using these diagrams you can also describe what happens if one variable is changed but the other is held constant.
For example, what happens to water at 1 atm, if the temperature increases from 25.0 degrees C to 75.0 degrees C? Nothing! It remains liquid! But, what happens if it goes from 25.0 degrees C to 105 degrees C? It changes from liquid to gas.
Also what if you had CO2 at 1 atm and -80 degrees C (it's a gas). However, if the pressure is increased to 5 atm, it changes from gas to solid!
*We can also use these graphs to find the melting and boiling point at a certain pressure.
For example, at 1 atm, the melting point of water is 0 degrees C and the boiling point of water is 100 degrees C.
*Finally, we can use phase change charts to point out the TRIPLE POINT. It's the point where at that given temperature and pressure, it is possible to have ALL 3 PHASES at once! For water, the triple point is at 0.006 atm of pressure, and 0.01 degrees C. For CO2, the triple point is at 5.1 atm of pressure and -56.7 degrees Celsius.
Objective 2.1.4: Infer simple calorimetic calculations based on the concepts of heat lost equals heat gained and specific heat.
In a closed system, energy can neither be created nor destroyed, only transferred (Law of Conservation of Energy).
Therefore, if we take one heated object and put it into another object, the heat should be transferred to a cooler object (Second Law of Thermodynamics). So any energy "lost" from the hot object should be "gained" by the cooler object. Because of that, we can set up a system of equations to solve for a mystery variable.
In class, we modeled this by heating a metal rod and dropping it into a beaker of water. We hypothesized that the temperature of the water would increase once the metal rod had been placed into it. We then used this information to calculate the mystery variable--how hot was the metal rod that we put into the water?
Sample Data:
46.0 gram Iron rod
Initial temperature as it sat on hot plate ????
Final temperature after it was dropped into water: 39.0 degrees C
Specific heat of Iron: 0.449 J/gC
50.0 mL Water = 50.0 grams Water
Initial temperature of water: 25.0 degrees C
Final temperature of water after iron was dropped in: 39.0 degrees C (the iron and water had the same final temp.)
Specific heat of Water: 4.18 J/gC
The only variables we are missing is the Ft of the Iron and the Q values for each of the substances. BUT--if the heat energy from the iron was released into the water, then the water absorbed it! Therefore, they are equal but opposite!
-Qiron = Qwater
-(m)(Tf-Ti)(Cp)iron = (m)(Tf-Ti)(Cp)water
-(46.0)(39.0 - Ti)(0.449) = (50.0)(39.0-25.0)(4.18)
-46.0(0.449)(39.0 - Ti) = 2926
-20.654(39.0 - Ti) = 2926
39.0 - Ti = -141.7
-Ti = -180.7
Ti = 180.7 = 181 degrees C *So, we heated the iron to 181 degrees C! That makes sense because it got really hot and sizzled when we placed it into the water!
In a closed system, energy can neither be created nor destroyed, only transferred (Law of Conservation of Energy).
Therefore, if we take one heated object and put it into another object, the heat should be transferred to a cooler object (Second Law of Thermodynamics). So any energy "lost" from the hot object should be "gained" by the cooler object. Because of that, we can set up a system of equations to solve for a mystery variable.
In class, we modeled this by heating a metal rod and dropping it into a beaker of water. We hypothesized that the temperature of the water would increase once the metal rod had been placed into it. We then used this information to calculate the mystery variable--how hot was the metal rod that we put into the water?
Sample Data:
46.0 gram Iron rod
Initial temperature as it sat on hot plate ????
Final temperature after it was dropped into water: 39.0 degrees C
Specific heat of Iron: 0.449 J/gC
50.0 mL Water = 50.0 grams Water
Initial temperature of water: 25.0 degrees C
Final temperature of water after iron was dropped in: 39.0 degrees C (the iron and water had the same final temp.)
Specific heat of Water: 4.18 J/gC
The only variables we are missing is the Ft of the Iron and the Q values for each of the substances. BUT--if the heat energy from the iron was released into the water, then the water absorbed it! Therefore, they are equal but opposite!
-Qiron = Qwater
-(m)(Tf-Ti)(Cp)iron = (m)(Tf-Ti)(Cp)water
-(46.0)(39.0 - Ti)(0.449) = (50.0)(39.0-25.0)(4.18)
-46.0(0.449)(39.0 - Ti) = 2926
-20.654(39.0 - Ti) = 2926
39.0 - Ti = -141.7
-Ti = -180.7
Ti = 180.7 = 181 degrees C *So, we heated the iron to 181 degrees C! That makes sense because it got really hot and sizzled when we placed it into the water!
Objective 2.1.5: Explain the relationships among pressure, temperature, volume, and quantity of gas, both qualitative and quantitative.
There are 5 general gas laws that are very necessary to be able to derive from information given within a problem: Boyle's Law, Charles' Law, Gay-Lussac's Law, The Combined Gas Law, and the Ideal Gas Law.
Boyle's Law: P1V1 = P2V2 [temperature is constant]
When pressure is increased, volume decreases (and vice versa).
Charles' Law: V1/T1 = V2/T2 [pressure is constant]
When temperature is increased, volume increases (and vice versa).
Gay-Lussac's Law: P1/T1 = P2/T2 [volume is constant]
When temperature is increased, pressure increases (and vice versa).
When pressure is increased, volume decreases (and vice versa).
Charles' Law: V1/T1 = V2/T2 [pressure is constant]
When temperature is increased, volume increases (and vice versa).
Gay-Lussac's Law: P1/T1 = P2/T2 [volume is constant]
When temperature is increased, pressure increases (and vice versa).
Combined Gas Law: P1V1/T1 = P2V2/T2 [nothing is constant!]
This law is used when you have many variables present (5) and are solving for one other piece. However, you can get all of the other "Name" laws by covering the variable that is constant in your problem. For example, if you are given info about temperature and pressure, then cover volume on both sides of the equal marks and you'll have the equation you need.
Ideal Gas Law: PV = nRT
The only gas law that doesn't look at a change in pressure, volume, or temperature. It focuses on how much substance (moles) are there given certain parameters of pressure, temperature, and volume.
This law is used when you have many variables present (5) and are solving for one other piece. However, you can get all of the other "Name" laws by covering the variable that is constant in your problem. For example, if you are given info about temperature and pressure, then cover volume on both sides of the equal marks and you'll have the equation you need.
Ideal Gas Law: PV = nRT
The only gas law that doesn't look at a change in pressure, volume, or temperature. It focuses on how much substance (moles) are there given certain parameters of pressure, temperature, and volume.
Upcoming Tests
Test #1:
Chapters 1-5
You should: know the difference between types of mixtures, elements and compounds; know how to convert measurements and calculate density; know how to perform percent error calculations, know about subatomic particles, their charges and how to find out how many are in each element isotope, or ion; know how to identify isotopes and calculate average atomic masses, know how to draw Bohr electron arrangements (the circles) and perform quantum electron configuration (the arrows); know about orbitals and which orbitals are filled by certain elements; know about atomic spectra and electron emissions and how to use the Bohr model of Hydrogen's spectrum to answer questions about the light emitted; know about the electromagnetic spectrum and how wavelength relates to frequency; know how the elements are arranged on the periodic table; know who created and later changed the periodic table; know the periodic trends and how to apply them (atomic radius, atomic number, ionization energy, electronegativity, reactivity, atom size v. ion size).
Chapters 1-5
You should: know the difference between types of mixtures, elements and compounds; know how to convert measurements and calculate density; know how to perform percent error calculations, know about subatomic particles, their charges and how to find out how many are in each element isotope, or ion; know how to identify isotopes and calculate average atomic masses, know how to draw Bohr electron arrangements (the circles) and perform quantum electron configuration (the arrows); know about orbitals and which orbitals are filled by certain elements; know about atomic spectra and electron emissions and how to use the Bohr model of Hydrogen's spectrum to answer questions about the light emitted; know about the electromagnetic spectrum and how wavelength relates to frequency; know how the elements are arranged on the periodic table; know who created and later changed the periodic table; know the periodic trends and how to apply them (atomic radius, atomic number, ionization energy, electronegativity, reactivity, atom size v. ion size).
Test #2:
Chapters 6-9
You should know: how to create electron dot structures for elements, how ions form and why, how to name ions, how to name ionic compounds, how to draw ionic compounds, how to draw covalent compounds, how to name covalent compounds, significance of metallic bonds, alloys, cations v. anions, oxidation numbers v. valence electrons, different types of covalent bonds and their strengths, VSEPR theory (shapes of covalent compounds) and why they are shaped the way they are, polar v. nonpolar bonds, weak bonds (dispersion, dipole, hydrogen), knowing what diatomic molecules are (and examples of some), network solids, and naming acids and bases.
Chapters 6-9
You should know: how to create electron dot structures for elements, how ions form and why, how to name ions, how to name ionic compounds, how to draw ionic compounds, how to draw covalent compounds, how to name covalent compounds, significance of metallic bonds, alloys, cations v. anions, oxidation numbers v. valence electrons, different types of covalent bonds and their strengths, VSEPR theory (shapes of covalent compounds) and why they are shaped the way they are, polar v. nonpolar bonds, weak bonds (dispersion, dipole, hydrogen), knowing what diatomic molecules are (and examples of some), network solids, and naming acids and bases.
Test #3:
Chapters 10-12
You should know: Significant Figures, Mole Calculations (mass to moles, moles to mass, mass to molecules, molecules to mass, atoms to mass, mass to atoms, volume to moles, moles to volume), Avogadro's Number and how to use it, The gas law constant (22.4 L/mole) and how to use it, Percent Composition, Empirical formulas v. Molecular formulas (and how to calculate them), how to read/write chemical equations, how to classify equations into categories (synthesis, decomposition, single replacement, double replacement, and combustion), writing net ionic equations, how to use solubility rules the predict the formation of a precipitate, how to balance an equation, and stoichiometry. Le Chatelier's principle, the factors that affect equilibrium, and how to calculate equilibrium constants, collision theory, how temperature, pressure and concentrations affect collisions, and why reactions occur, the difference between endothermic and exothermic reactions and what happens to energy in these reactions, .
Chapters 10-12
You should know: Significant Figures, Mole Calculations (mass to moles, moles to mass, mass to molecules, molecules to mass, atoms to mass, mass to atoms, volume to moles, moles to volume), Avogadro's Number and how to use it, The gas law constant (22.4 L/mole) and how to use it, Percent Composition, Empirical formulas v. Molecular formulas (and how to calculate them), how to read/write chemical equations, how to classify equations into categories (synthesis, decomposition, single replacement, double replacement, and combustion), writing net ionic equations, how to use solubility rules the predict the formation of a precipitate, how to balance an equation, and stoichiometry. Le Chatelier's principle, the factors that affect equilibrium, and how to calculate equilibrium constants, collision theory, how temperature, pressure and concentrations affect collisions, and why reactions occur, the difference between endothermic and exothermic reactions and what happens to energy in these reactions, .
You should know how to balance equations showing the nuclear decay of atoms (alpha, beta, gamma, etc...) How to calculate half-lives of radioactive elements. Contrast fission with fusion (nuclear chemistry).
Test #4:
You should know: what occurs when phases (states) change, how to read heating/cooling curves, how to read phase-change diagrams and how temperature and pressure affect the phase of a substance, the law of conservation of energy (1st Law of Thermodynamics), the law of entropy (2nd Law of Thermodynamics), how to use gas laws (Boyles, Charles, Gay-Lussac, Combined Gas Law, Ideal Gas Law) to calculate pressures, volumes, temperatures, and moles of a gas. You should also know about the types of solutions and how they form, how to calculate molarity, how to calculate dilutions, characteristics of acids/bases, pH scale, and how to perform titrations to find the molarity of a mystery acid.
Test #4:
You should know: what occurs when phases (states) change, how to read heating/cooling curves, how to read phase-change diagrams and how temperature and pressure affect the phase of a substance, the law of conservation of energy (1st Law of Thermodynamics), the law of entropy (2nd Law of Thermodynamics), how to use gas laws (Boyles, Charles, Gay-Lussac, Combined Gas Law, Ideal Gas Law) to calculate pressures, volumes, temperatures, and moles of a gas. You should also know about the types of solutions and how they form, how to calculate molarity, how to calculate dilutions, characteristics of acids/bases, pH scale, and how to perform titrations to find the molarity of a mystery acid.
Final Exam Practice!
| fall_2021_chemistry_final_a.docx | |
| File Size: | 240 kb |
| File Type: | docx |
We went over the exam above in class! The exam below is another version that you can try for practice if you'd like!
| fall_2021_chemistry_final_b.docx | |
| File Size: | 240 kb |
| File Type: | docx |